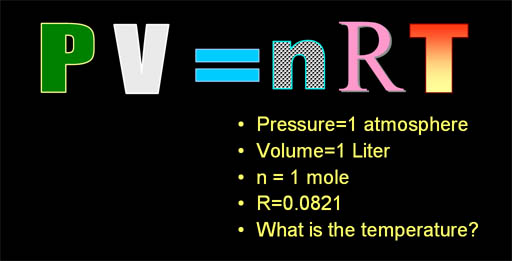Unit 11: Gases
Essential Questions
- What is absolute zero?
- How does the kinetic molecular theory explain the behavior of gases?
- How are pressure units converted?
- What is Daltons Law of partial pressures?
- Why are they called Laws and not theories?
- How are temperature, pressure and volume related?
- What is Standard temperature and pressure?
- How can the volume, pressure or temperature of a gas be calculated?
- Can the amount of moles of a gas be related to the pressure, volume and temperature of the sample?
- Do gases always follow the idea gas law?
- How can balanced equations be used to calculate the volume of gases formed in chemical reactions?
- What is absolute zero?
Vocabulary
- Polar
covalent bond
- dispersion force
- dipole-dipole force
- hydrogen bond
- viscosity
- surface tension
- melting point
- vaporization
- evaporation
- vapor pressure
- boiling point
- freezing point
- condensation
- deposition
Standards
SC.912.P.12.10 Interpret the behavior of ideal gases in terms of kinetic molecular theory.
- Students will understand that all movement of particles inside of a substance stopped when absolute zero is reached.
- Student identifies the kinetic-molecular theory explains the behavior of gases. It assumes gases:
- Have small particles with large, empty spaces between them.
- Gas particles are in constant, random motion.
- Gas particles have elastic collisions.
- Temperature is a measure of the average kinetic energy of the gas particles.
- Gases exert pressure on a container when the gas particles collide with the walls of the container.
- Gas pressure is measured with barometers and pressure gauges.
- The SI unit of pressure is the Pascal. Atmospheres are also used to measure pressure (1 atm = 101 kPa).
- Students will convert between pressure conversions of atm, kPa, and mmHg. (1 atm = 760.0 mmHg = 101.325 kPa will be provided.)
- Honors Extension: The pressure of a mixture of gases is equal to the sum of the pressures of the individual components (Dalton’s Law).
- Students calculate the pressure of a gas mixture from the pressures of the components.
SC.912.N.3.3 Explain that scientific laws are descriptions of specific relationships under given conditions in nature, but do not offer explanations for those relationships.
- Student applies the gas laws to amounts of gas to predict changes in the pressure, temperature and volume.
- Boyle’s Law
- Charles’s Law
- Gay-Lussac’s Law
- STP is one atmosphere pressure (101.3 kPa) and 0oC.
- Honors Extension: Students relate the pressure, volume, temperature and moles of gas to each other using the ideal gas law.
- Students can choose the correct value of R to use in their calculations.
- Students recognize the conditions where the ideal gas law cannot be used (High pressure, Low temperature)
- Honors Extension: Student uses the gas laws to calculate the volume, pressure and temperature of gases.
- The combined gas law
- STP is one atmosphere pressure (101.3 kPa) and 0oC.
- C.912.P.8.9 Apply the mole concept and the law of conservation of mass to calculate quantities of chemicals participating in reactions
- Students use the coefficients of a balanced equation to perform stoichiometric volume to volume calculations involving gases.
- Students relate the number of particles of a gas to the volume of the gas at constant pressure, temperature and volume.
- 1 mol = 22.4 L
Textbook
READ CHAPTER 13: PAGES 440-464