Unit 10: States of Matter
Essential Questions
- What is the relationship between temperature and kinetic energy?
- How are degrees Celsius converting into Kelvin?
- What are some examples of chemical properties? Physical properties?
- What are some examples of some chemical changes? Physical changes?
- How do intermolecular forces determine a substances state?
- How do intermolecular forces determine the range of motion of particles in solids and liquids?
- How do intermolecular forces determine the range of motion of particles in gases?
- How are plasmas different from gases?
- What makes matter change phase?
- How does a phase change reach dynamic equilibrium?
- (Optional) How can scientists summarize phase changes in a graph?
- What is the relationship between temperature and kinetic energy?
Vocabulary
- Polar
covalent bond
- dispersion force
- dipole-dipole force
- hydrogen bond
- viscosity
- surface tension
- melting point
- vaporization
- evaporation
- vapor pressure
- boiling point
- freezing point
- condensation
- deposition
Standards
SC.912.P.10.5 Relate temperature to the average molecular kinetic energy.
- Students know that temperature and average kinetic energy are directly related.
- Students will convert temperature units from Kelvin to degrees Celsius and vice versa. (K = C +273 will be provided.)
- Student will identify the differences between chemical and physical properties.
- Physical: states of matter, color, texture, mass, density
- Chemical: reactivity with acids/bases, flammability, reactivity with oxygen
- Student will identify the differences between chemical and physical changes.
- Intermolecular and intramolecular forces determine a substances state at a given temperature.
- Students identify the three intermolecular forces and know which is strongest.
- Dispersion forces.
- Dipole-dipole forces
- Hydrogen bonds
- Students identify the properties of gases, liquids and solids:
- Particle arrangement
- Compressibility
- Fluidity
- Students will define and give an example of plasma.
- Molecular energy
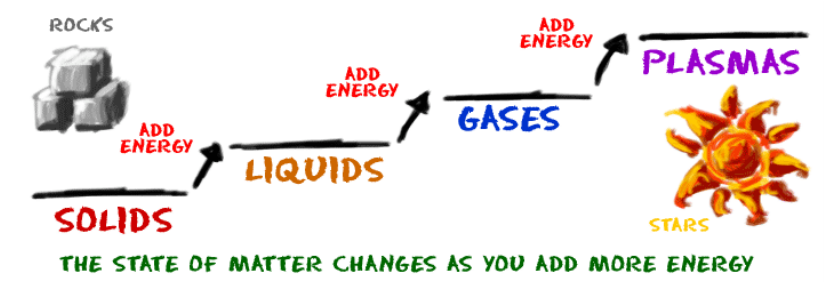
- The addition of energy causes endothermic phase changes.
- Melting
- Vaporization
- Sublimation
- The removal of energy causes exothermic phase changes.
- Freezing
- Condensation
- Deposition
- Intermolecular forces determine the phase of a material.
- Students will know that the rate of vaporization will equal the rate of condensation in a sealed container.
- Optional Extension (not assessed): Phase diagrams summarize phase changes.
- Students can read a phase diagram and identify:
- Normal freezing point
- Normal boiling point
- The triple point
- The critical point
Textbook
READ CHAPTER 12: PAGES 401- 430