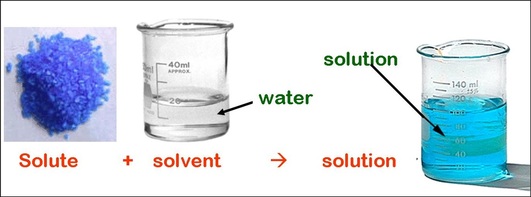
Unit 12: Solutions
Essential Questions
- What factors affect the formation of solutions?
- How is concentration of a solution calculated using molarity?
- How does a solution become saturated or supersaturated?
- What factors affect the solubility of gases and solids in liquids?
- What's the difference between strong, weak, and nonelectrolytes?
- What properties of solutions are colligative properties?
- How does an increase in vapor pressure affect the colligative properties of a solution?
- How would an equation be written for the dissociation of ionic solutes in water?
- Why does oil not dissolve in water according to the factors that affect solubility?
- How does an increase in temperature and pressure of a solution affect the solubility of the solute?
- How does polarity of the solute and solvent affect the solubility of a solution?
- Compare, contrast, and provide examples of suspensions, colloids, and solutions similar and different.
- What does a solubility curve represent?
- What factors affect the formation of solutions?
Vocabulary
· Kinetic molecular theory
· elastic collision
· temperature
· pressure
· absolute zero
· combined gas law
· molar volume
· ideal gas law
· ideal gas constant
Standards
SC.912.L.18.12 Discuss the special properties of water that contribute to Earth's suitability as an environment for life: cohesive behavior, ability to moderate temperature, expansion upon freezing, and versatility as a solvent
- Students will understand that concentration refers to the ratio of the amount of solute to solvent in the solution and that molarity is a common concentration used in the chemistry lab.
- Students will solve molarity problems finding molarity, moles or liters given the other two variables.
- Students will calculate the mass of a solvent needed to prepare a solution. Students will calculate the molarity of a solution given the mass of the solute and the volume of the solution.
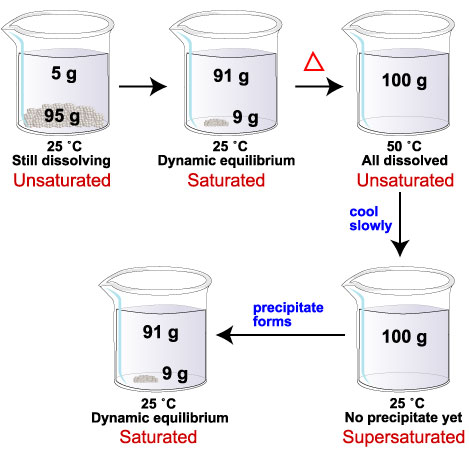
- Students will define and identify solutions that are unsaturated, saturated, and supersaturated.
- Students will identify factors that affect solubility, specifically temperature, pressure (for gases), and polarity of solute and solvent. “Like dissolves like.”
- Students will list the three factors (stirring, surface area, and temperature) that affect the rate of dissolving of a solid solute in water and explain the role of each.
- Students will identify a solution as an electrolyte (and determine if it's a strong or weak electrolyte) or nonelectrolyte.
- Students understands vapor pressure and its relationship to the other colligative properties. Student can state several practical applications of colligative properties of aqueous solutions.
- Students will be able to write equations for the dissociation of ionic solutes in water.
- Students will be able to solve molality problems.
- Students will be able to solve freezing point change and boiling point change problems for aqueous solutions.
- Student will understand the impacts of oil spills (such as the 2010 Gulf of of Mexico oil spill) on environmental quality.
- Student will identify factors that affect solubility, specifically temperature, pressure (for gases), and polarity of solute and solvent. “Like dissolves like.”
- Student will understand the impacts of oil spills (such as the 2010 Gulf of of Mexico oil spill) on environmental quality.
- Student will identify common suspensions, colloids, and solutions.
- Student will interpret solubility curves.
- Student will identify solutions that are unsaturated, saturated, and supersaturated using solubility curves.
Textbook
READ CHAPTER